In recent years, the momentum of technological revolution and industrial transformation has been accelerating. Robotics, as a major innovation integrating information technology, control systems, and intelligent manufacturing, is increasingly pivotal in this transformation, often hailed as the "crown jewel" of manufacturing.
Sidebar
Knowledge base

IDTechEx Explores 3D Electronics and Additive Electronics in the Automotive Industry
With 3D electronics, bulky PCBs can be replaced, resulting in sleek and integrated designs. The automotive industry is one of the largest sectors benefiting from this technology, as electronics can be printed in a thin layer onto surfaces or integrated within components, which can be particularly useful for human-machine interfaces (HMIs).

BLT: Metal AM Printing's Shift from Technology-Driven to Cost-Driven
"Additive manufacturing isn’t just about comparing build volume sizes across different manufacturers. While build volume is a technical consideration, it's only part of the bigger picture. To truly penetrate the industrial sector and usher in the era of mass production, our products must be manufacturable and affordable for customers.

Increasing efficiency in on demand 3d printing
The past few years have been challenging, with a number of crises, such as supply bottlenecks and geopolitical conflicts. As a technology company in a highly competitive market environment, we have faced these head-on. We have not been spared, but we have achieved the best consolidated result in our company’s history in the 2023 financial year.

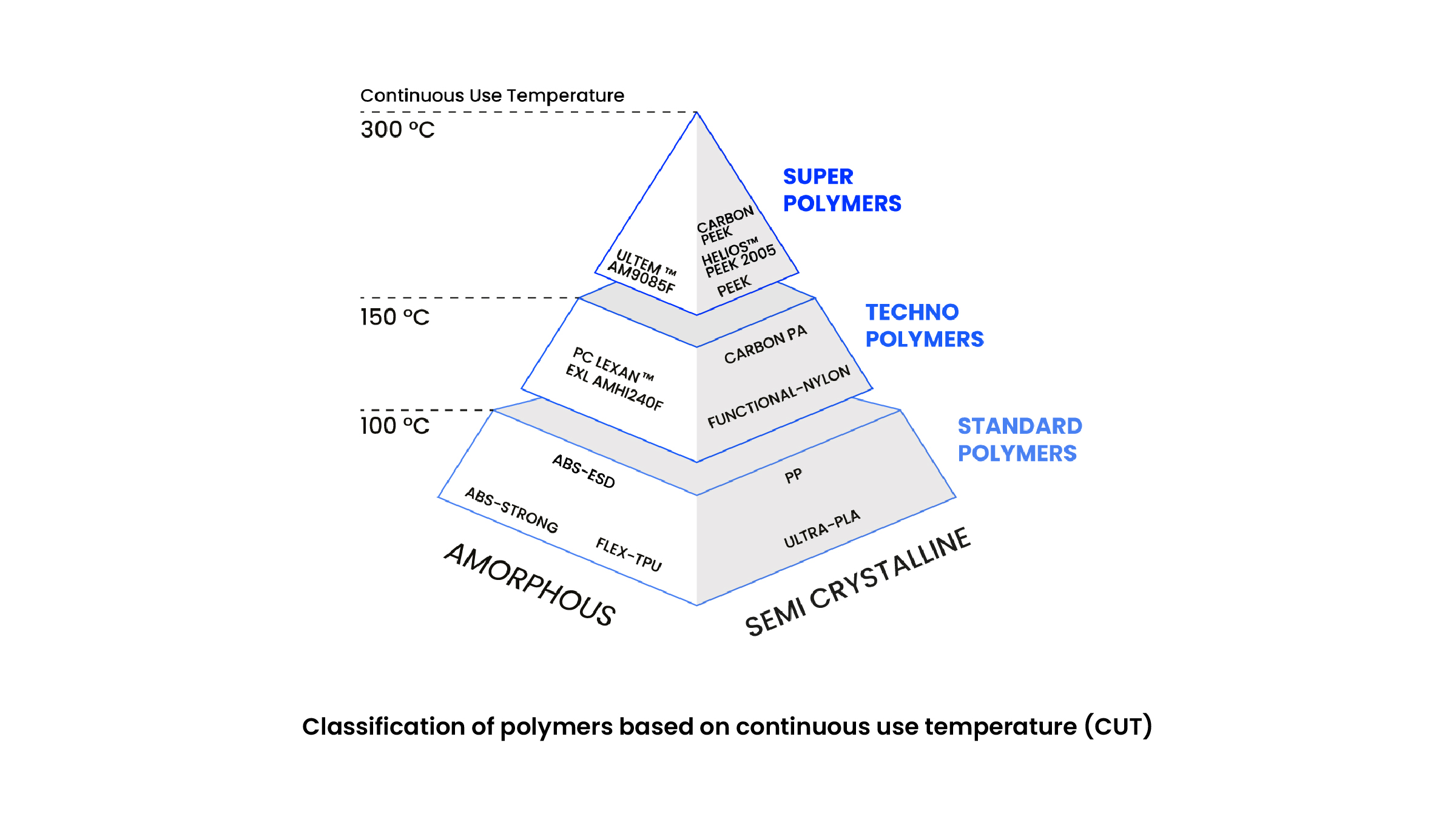
The importance of the ARGO 1000 HYPERMELT's heated chamber for large format super polymer printing
3D printing has revolutionized the manufacturing industry, enabling the production of complex components with unprecedented ease and versatility. However, when it comes to super polymers and large format parts, the challenge becomes more complex. Roboze's ARGO 1000 HYPERMELT technology is here to overcome these challenges and offer an advanced solution.
Roboze: Benefits and advantages of 3D printing
What you should know about industrial 3D printing: advantages and disadvantages
Additive manufacturing (AM), also known as 3D printing, is one of the fastest developing manufacturing technologies in the world. It is a process of layer-by-layer material deposition to produce parts from 3D models. 3D printing has demonstrated its efficacy especially in low-volume, high-customisation industries such as aerospace, and there are many reasons for this.

4 Metal Additive Manufacturing Myths Debunked: Laser Powder Bed Fusion (LPBF)
By Evan Butler-Jones, Vice-President, Product & Strategy, Equispheres
In the rapidly evolving world of metal additive manufacturing, laser powder bed fusion has a bit more history behind it than some of the newer techniques. L-PBF technologies are advancing by leaps and bounds but a few outdated myths about powder-based processes persist. I’ve tried to dispel a few of the common myths below.
Addressing the Global Kidney Shortage: Can 3D Printed Organs Be the Solution?
Rising kidney transplant demands underscore a pressing global health concern. While 3D-printed organs promise a potential solution, challenges in printing speed and accuracy persist. Leading the way, Vidmantas Šakalys of Vital 3D provides valuable insights and hints at a transformative solution on the horizon.
New Report from QAD Redzone Identifies $1.4M Annual Labor Savings Across 1,000 Plants
The fifth biennial Productivity Benchmark Report analyzed results from 1,000 plants over a 10-year period.

The critical role of 3D printing support material
By Infinite Editorial Team
Get to know your supporting partner in additive manufacturing
Unless your goals are limited to custom hockey pucks and little gnome figures, support materials will play a critical role in your 3D printing efforts. 3D printing support materials enable you to create and print complex geometries, parts with internal features, and parts that cannot be manufactured by any other method. Without support, your build material would deform before it hardened.

Custom plastics manufacturing services - Find the best injection molding professionals.
Functionality and flawless design are always important for manufacturing every product. However, to ensure this flawlessness, you must invest in plastic injection molding projects. You will get custom plastic manufacturing solutions from injection molding companies.

 Deutsch (Germany)
Deutsch (Germany)  Polski (PL)
Polski (PL) 






